PrEP and PEP for HIV: Before and After Prevention F O U RT H A N N U A
47 Slides2.98 MB
PrEP and PEP for HIV: Before and After Prevention F O U RT H A N N U A L U SC C O L L E G E O F N U R S I N G CLINICAL PRACTICE CONFERENCE S A B R A S . C U S T E R , D N P, M S , F N P - B C C L I N I C A L A S S O C I AT E P R O F E S S O R , C O L L E G E O F N U R S I N G P R E P S L I D E S O R I G I N A L LY B Y D I V YA A H U J A , M D A S S O C I AT E P R O F E S S O R , S C H O O L O F M E D I C I N E
Annual HIV Incidence There are approximately 50,000 new infections in the US each year Homosexual men (MSMs), particularly young, African- American MSMs are disproportionately affected African-Americans in general are disproportionately affected http://www.cdc.gov/hiv/statistics/overview/ataglance.html
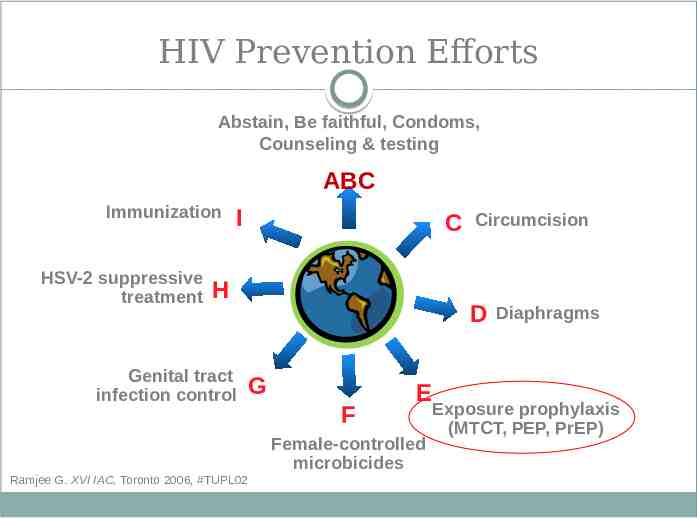
HIV Prevention Efforts Abstain, Be faithful, Condoms, Counseling & testing ABC Immunization HSV-2 suppressive treatment I C H Genital tract infection control Ramjee G. XVI IAC, Toronto 2006, #TUPL02 Circumcision D G F E Female-controlled microbicides Diaphragms Exposure prophylaxis (MTCT, PEP, PrEP)
PrEP P R E - E X P O SU R E P R OP H Y L A X I S
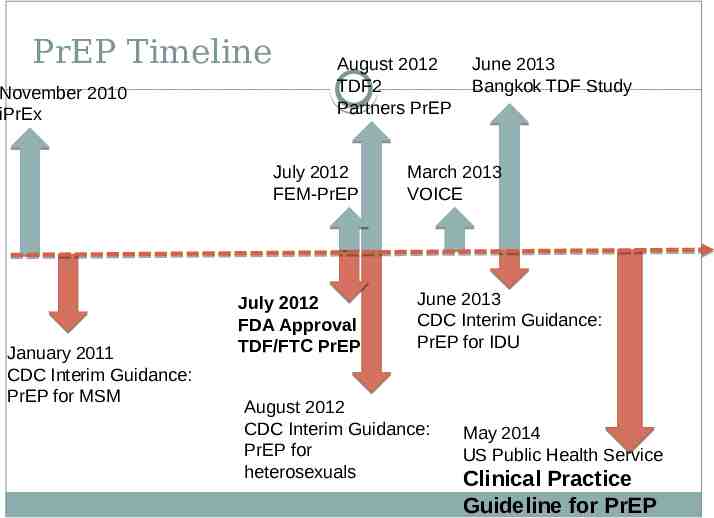
PrEP Timeline November 2010 iPrEx August 2012 TDF2 Partners PrEP July 2012 FEM-PrEP January 2011 CDC Interim Guidance: PrEP for MSM July 2012 FDA Approval TDF/FTC PrEP June 2013 Bangkok TDF Study March 2013 VOICE June 2013 CDC Interim Guidance: PrEP for IDU August 2012 CDC Interim Guidance: PrEP for heterosexuals May 2014 US Public Health Service Clinical Practice Guideline for PrEP
Barriers to Use of PrEP Eligibility Adherence Increased risky sexual practices Side effects Reimbursement Patient accountability Provider knowledge, comfort, and willingness to prescribe
PrEP Candidates Men who have sex with men (MSM) who: Have an HIV-positive sexual partner Have a recent bacterial STI Have a high number of sex partners Have a history of inconsistent/no condom use Engage in commercial sex work Transgender individuals who: Engage in high-risk sexual behaviors http://www.cdc.gov/hiv/pdf/guidelines/PrEPguidelines2014.pdf
PrEP Candidates Heterosexual women and men who: Have an HIV-positive sexual partner Have a recent bacterial STI Have a high number of sex partners Have a history of inconsistent/no condom use Engage in commercial sex work Live in a high-prevalence area or network Injection drug users (IDU) who: Have an HIV-positive injecting partner Share injection equipment Have been through recent drug treatment (but currently injecting) http://www.cdc.gov/hiv/pdf/guidelines/PrEPguidelines2014.pdf
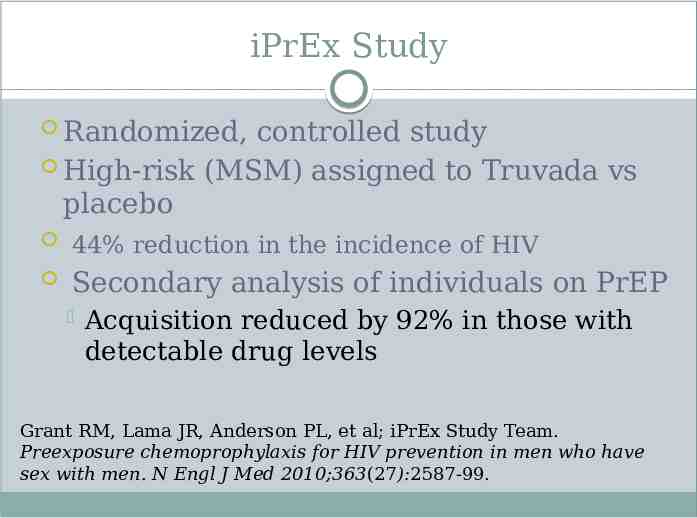
iPrEx Study Randomized, controlled study High-risk (MSM) assigned to Truvada vs placebo 44% reduction in the incidence of HIV Secondary analysis of individuals on PrEP Acquisition reduced by 92% in those with detectable drug levels Grant RM, Lama JR, Anderson PL, et al; iPrEx Study Team. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med 2010;363(27):2587-99.
The PROUD Study The PROUD study enrolled MSMs from 13 sexual health clinics in England between 2012 and 2014 Eligibility criteria : negative HIV test condomless anal intercourse in the previous 90 days 545 MSM randomized 1:1 to daily TDF/FTC Either immediately (IMM) Or after a deferral (DEF) period of 12 months Relative reduction in HIV acquisition of 86% in the Immediate arm (62-96%; P 0.0002). Confirmed STI (rectal chlamydia/gonorrhea) in Immediate arm-29% Deferred arm -27% McCormack S, Dunn D, Desai M. (2016) Pre-exposure prophylaxis to prevent the acquisition of HIV-1 infection (PROUD) The Lancet 387(10013),53-60.
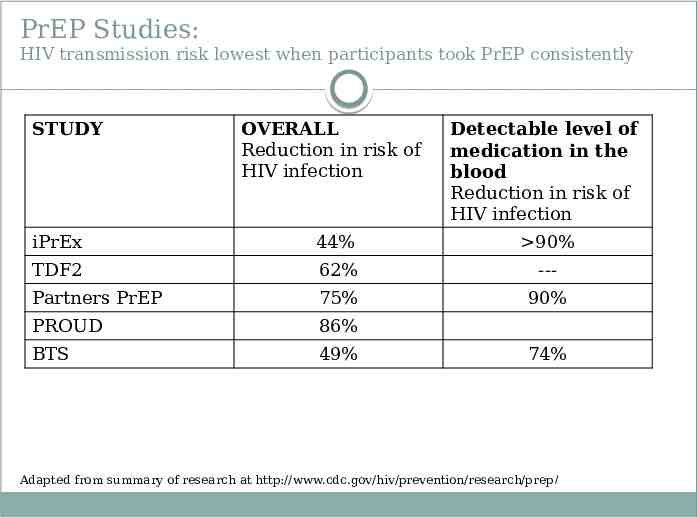
PrEP Studies: HIV transmission risk lowest when participants took PrEP consistently STUDY OVERALL Reduction in risk of HIV infection Detectable level of medication in the blood Reduction in risk of HIV infection iPrEx 44% 90% TDF2 62% --- Partners PrEP 75% 90% PROUD 86% BTS 49% 74% Adapted from summary of research at http://www.cdc.gov/hiv/prevention/research/prep/
Rule out Acute HIV Infection before PrEP Symptoms of Acute HIV Fever Fatigue Myalgia Skin rash Headache Pharyngitis Cervical Lymphadenopathy Arthralgia Night sweats Diarrhea Daar ES, Pilcher CD, Hecht FM. Curr Opin HIV AIDS. 2008;3(1):10-15.
Case Study 32 year old black female seeks pregnancy. Tested six months ago and is HIV-negative. Her male partner is HIV-positive and not currently on antiretroviral treatment. What do you recommend?
PrEP for Safe Conception Mother takes PrEP to prevent her from acquiring HIV from male partner Limited data In the small studies, no HIV transmission to the woman Among women in Antiretroviral Pregnancy Registry: no birth defects with Truvada
PrEP for Serodiscordant Partners 54 year male Monogamous with HIV-positive partner The HIV positive partner has had a undetectable viral load for 1 year. Would you give PrEP to the HIV negative partner?
CDC guidelines support PrEP even when the HIV positive partner is undetectable Likelihood of acquiring HIV is very low from a virally-suppressed HIV-positive partner However: Partner may have virological blips Partner may become non adherent Patient may not be monogamous
2014 PrEP Prescribing Guidelines Determine eligibility: negative HIV test, high risk of infection and creatinine clearance 60ml/min Assess for Hepatitis B sAg and pregnancy (female patients) Prescribe : Tenofovir-emtricitabine (Truvada) one pill once daily (90 day supply) Monitor: creatinine clearance, HIV status, and pregnancy every 3 months and STI screen every 6 months; counsel on adherence http://www.cdc.gov/hiv/pdf/guidelines/PrEPguidelines2014.pdf and risk reduction
Providing PrEP Before starting PrEP: Clinical eligibility Educate Side effects Limitations Daily adherence Symptoms of seroconversion Monitoring schedule Safety Criteria for discontinuation Partner information Social history: housing, substance use, mental health, domestic violence
Providing PrEP After confirmation of clinical eligibility: Prescribe no more than 90-day supply of PrEP Truvada 1 tablet PO daily (tenofovir 300mg emtricitabine 200mg) Insurance prior approval Truvada for PrEP Medication Assistance Program
Providing PrEP 3-month visit: HIV test Assess for acute infection Check for side effects Pregnancy testing Prescribe 90-day supply of medication Every visit: Assess adherence Risk reduction counseling Provide condoms
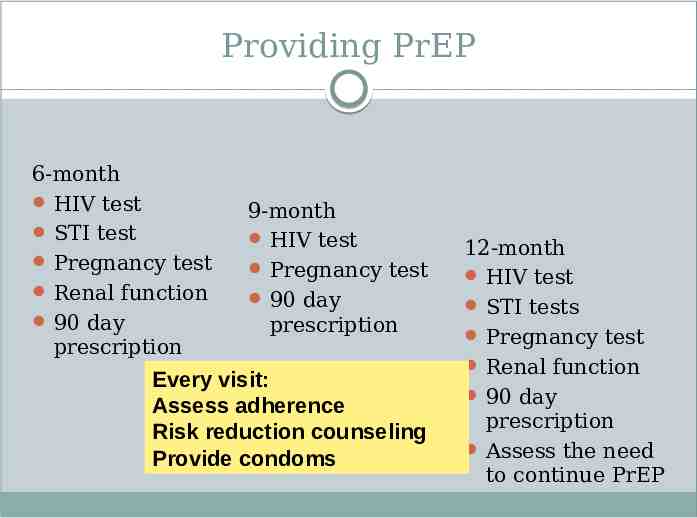
Providing PrEP 6-month HIV test STI test Pregnancy test Renal function 90 day prescription 9-month HIV test Pregnancy test 90 day prescription Every visit: Assess adherence Risk reduction counseling Provide condoms 12-month HIV test STI tests Pregnancy test Renal function 90 day prescription Assess the need to continue PrEP
Discontinuing PrEP Positive HIV result Acute HIV signs or symptoms Non-adherence Renal disease Changed life situation: lower HIV risk http://www.cdc.gov/hiv/pdf/guidelines/PrEPguidelines2014.pdf
On-Demand PrEP IPERGAY: randomized trial, 400 high-risk MSM peri-coital PrEP: Truvada 4 tablets or placebo two 2 to 24 hours before sex, a second dose 24 hours later, and a last one 24 hours later 86% reduction in HIV acquisition with on-demand PrEP
Future of PrEP Nano-formulations or long acting meds Cabotegravir: investigational HIV integrase inhibitor Can be administered orally or as long-acting subcutaneous or intramuscular injection Single injection of long-acting version could be effective for up to 3 months
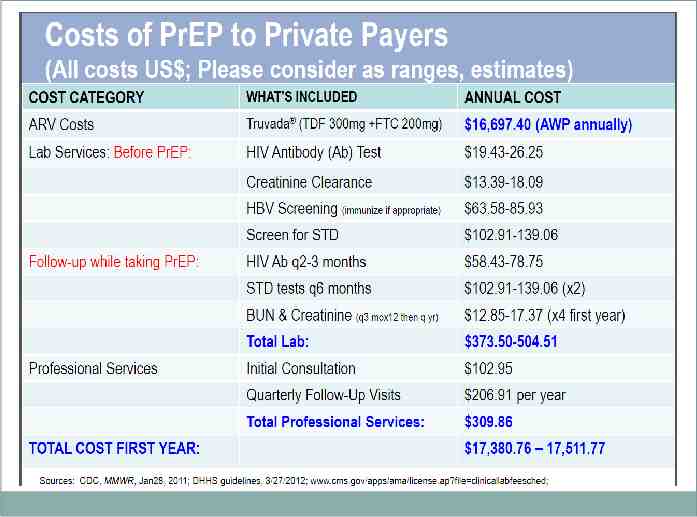
Financial Issues Coverage for PrEP varies in US Most private insurers are providing coverage, with prior authorization requirements Potential issue of economic disparity for uninsured/low-income patients
Reimbursement for PrEP Follow the CDC Clinical Guidelines for PrEP Common ICD-10 codes for PrEP counseling: Z20.2 “Contact with and (suspected) exposure to infections with a predominantly sexual mode of transmission” Z11.4 “Encounter for screening for HIV” Z11.3 “Encounter for screening for infections with a predominantly sexual mode of transmission” Use the usual E/M charge based on length/complexity of visit
Reimbursement for PrEP Although regular HIV screening labs are rated “A” by USPSTF and should be covered without a “patient due balance”, the greater frequency of labs needed while on PrEP may generate charges Broad coverage by Medicaid for PrEP – prior authorizations may be necessary -SC Medicaid does provide Truvada for PrEP with NO prior authorization necessary Broad coverage for Truvada by private insurers as well, prior authorizations also likely necessary
Truvada Costs Out-of-pocket estimated expense: 1,300 a month The manufacturer of Truvada offers assistance to uninsured individuals: http:// www.gilead.com/responsibility/us-patient-acc ess/truvada%20for%20prep%20medication% 20assistance%20programstar The manufacturer of Truvada also has a copay assistance program applicable to some insured individuals: https://start.truvada.com/
Payment Assistance Information Project Inform: http:// www.projectinform.org/pdf/PrEP Flow Chart.pdf -explains payment assistance for insured and un-insured Assessment of Medicaid Coverage of HIV/AIDS Prevention, Screening, and Care Services: A Ten State Review: https:// careacttarget.org/sites/default/files/supporting-files/Ass essment%20of%20Medicaid%20Coverage.pdf
PrEP works. Lets use it appropriately and responsibly
PEP P O ST- EX P O S U R E P R O P H Y L AX I S
PEP Categories oPEP –for occupational exposures HCWs who may experience a cut, needle stick, or other potentially infectious body fluid exposure “on the job” nPEP –for non-occupational exposures Persons who are potentially exposed to HIV through consensual or forced intercourse, accidental puncture wounds, or IVDU
Occupational HIV Exposures Definition of exposure: percutaneous injury or contact of mucous membrane or non-intact skin with blood, tissue, or other potentially infectious body fluids -potentially infectious body fluids: semen, vaginal secretions, CSF, synovial fluid, pleural fluid, peritoneal fluid, pericardial fluid, amniotic fluid -only potentially infectious if visibly bloody: feces, nasal secretions, saliva, sputum, sweat, tears, urine, vomitus
Risk of Occupational HIV Transmission Average risk after percutaneous exposure to HIV-infected blood: 0.3% Average risk after mucous membrane exposure to HIV-infected blood: 0.09% Factors that increase risk of HIV transmission: -device (needle, etc) is visibly contaminated with blood -needle had been placed directly into a vein or artery -deeper injuries -amount of HIV present in the source patient’s blood
Non-Occupational HIV Exposures Sexual contact, consensual or forced Accidental cuts or punctures with sharp objects Intentional use of contaminated or shared needles for IVDU
Evaluation of Non-Occupational Exposures HIV status of the potentially exposed person -baseline rapid testing should be conducted to ensure they are not already HIV-positive Timing and frequency of exposure -nPEP should be initiated within 72 hours of exposure Risk of HIV acquisition based on type of exposure HIV status of the exposure source -often difficult to obtain for non-occupational exposures
Risk of Non-Occupational HIV Transmission Receptive anal intercourse 1.38% Receptive penile-vaginal intercourse 0.08% Needle sharing for IVDU 0.63% Needlesticks 0.23% As with occupational exposures, increased amount of HIV present in the source patient’s blood or body fluids increases risk of transmission For sexual exposures, non-intact mucous membranes increases risk of transmission
Other Considerations for Possible Sexual Exposures Prophylaxis for bacterial STIs, trichomoniasis Testing for Hepatitis B and C Pregnancy prevention for female patients Counseling and other support for survivors of sexual assault
Shared Principles for All Types of PEP Importance of quick initiation of PEP following possible HIV exposure Importance of HIV tests for the potentially exposed patient Use of a “complete” three-drug regimen for PEP Duration of treatment is 28 days Follow-up testing required at 6 weeks and 4 months (with newest, 4th-generation Ag/Ab tests)
Time is of the Essence! PEP should be initiated as quickly as possible for all types of exposures nPEP guidelines state effectiveness is unlikely 72 hours after exposure For frequent possible exposures, discuss PrEP with the patient
Baseline Testing for Exposed Person Gold standard is the 4th generation Ag/Ab test to establish that exposed person is currently HIV-negative -decreased window period compared to older tests Familiarity with common manifestations of acute HIV infection could be helpful for rare instances of very acute HIV exposed person -can proceed based on 4th generation test results, but consult to ID would be prudent
What to Prescribe? oPEP and nPEP guidelines recommend the same regimen: emtricitabine/tenofovir DF (Truvada)200/300 mg daily raltegravir (Isentress) 400 mg twice daily For 28 days
Alternative Choices Dolutegravir (Tivicay) 50 mg can be substituted for raltegravir (Isentress), advantage is once-daily dosing For renal dysfunction (creatinine clearance 59 mL/min), can substitute zidovudine/lamivudine 300/150 (Combivir) or dose-adjust the individual components nPEP guidelines include recommendations for weight-adjustment for children
Additional Items Guidelines recommend checking serum creatinine at 4-6 weeks after exposure (along with first follow-up HIV test) for patients prescribed Truvada Provision of ‘starter packs’ in the emergency department setting is recommended for survivors of sexual assault who need PEP Consider follow-up and broader support needs for survivors of sexual assault
References US Public Health Service (2014). Preexposure Prophylaxis for the Prevention of HIV Infection in the United States – 2014. Retrieved from: http://www.cdc.gov/hiv/pdf/guidelines/PrEPguidelines2014.pdf Kuhar, D, et al (2013, Sept). Updated US Public Health Service Guidelines for the Management of Occupational Exposures to Human Immunodeficiency Virus and Recommendations for Postexposure Prophylaxis. Infection Control and Hospital Epidemiology, (34,9), pp.875-892 CDC, US DHHS (2016). Updated Guidelines for Antiretroviral Postexposure Prophylaxis After Sexual, Injection Drug Use, or Other Nonoccupational Exposure to HIV – United States, 2016. Retrieved from: https://stacks.cdc.gov/view/cdc/38856