National Standard for December 2015 WA Health
53 Slides4.77 MB
National Standard for December 2015 WA Health
Presentation Summary Labelling for safety Labelling Standard Aims Minimum requirements Outline and content Application in clinical practice User-applied labelling of injectable medicines 2
Labelling for Safety Labelling of injectable medicines, fluids and delivery devices is a major patient safety issue Labelling is often not done or incomplete, omitting information such as: name of medicine medicine dose patient name time of preparation. Incomplete/omitted labelling is a source of medication error User-applied labelling of injectable medicines 3
Medicine administration errors Medicine administration errors related to absent or inadequate labelling include: Wrong medicine Wrong route Wrong patient Labelling errors are particularly associated with: Patient transfer between clinical areas Perioperative sterile field 0.9% sodium chloride flush Line misconnections User-applied labelling of injectable medicines 4
Medicine administration errors Case Report 1 10mg morphine was given in error as the clinician thought the syringe contained 0.9% sodium chloride. The unlabelled syringe had a 0.9% sodium chloride ampoule attached. (unpublished) User-applied labelling of injectable medicines 5
Medicine administration errors Case Report 2 A patient was given intravenous (IV) lignocaine with adrenaline solution intended for local anaesthetic infiltration. This syringe had been drawn up and placed in a kidney dish alongside IV morphine and midazolam for procedural sedation. (unpublished) User-applied labelling of injectable medicines 6
The Labelling Standard Draft recommendations were developed by NSW Therapeutic Advisory Group Safer Medicines Group National consultation and pilot testing supported by the Australian Commission on Safety and Quality in Health Care commenced in 2009 Labelling Recommendations endorsed by Australian Health Ministers in November 2010 Further evaluation, particularly in perioperative areas and interventional procedure rooms Version 2 released February 2012 National Standard released September 2015 User-applied labelling of injectable medicines 7
The Labelling Standard A national standard for clinical practice in Australia Identifies medicines and fluids removed from manufacturer’s original packaging prior to patient administration Identifies line route User-applied labelling of injectable medicines 8
Labelling Standard Aims Provide standardisation for user-applied labelling of injectable medicines Provide minimum requirements for userapplied labelling of injectable medicines Promote safer use of injectable medicines User-applied labelling of injectable medicines 9
Labelling Standard Development Based on: International literature/recommendations Australian Standard AS4940: 2002 User-applied identification labels for use on fluid bags, syringes and drug administration lines. International Standard ISO 26825:2008 Anaesthetic and respiratory equipment – user-applied labels for syringes containing drugs used during anaesthesia – colours, design and performance Expert opinion Pilot testing Reported medicine administration incidents User-applied labelling of injectable medicines 10
Labelling Standard Minimum requirements All medicines and fluids removed from the manufacturer’s or hospital pharmacy’s original packaging must be identifiable. All containers (e.g. bags, syringes) containing medicines leaving the hands of the person preparing the medicine must be labelled. Prepare and label one medicine at a time before the preparation and labelling of a subsequent medicine. Any medicine or fluid that cannot be identified (e.g. in an unlabelled syringe or other container) is considered unsafe and should be discarded. User-applied labelling of injectable medicines 11
Labelling Standard Consultation User-applied labelling of injectable medicines 12
Labelling Standard Outline What should be labelled What should be included on the label Where the label should be placed Where the Labelling Standard applies User-applied labelling of injectable medicines 13
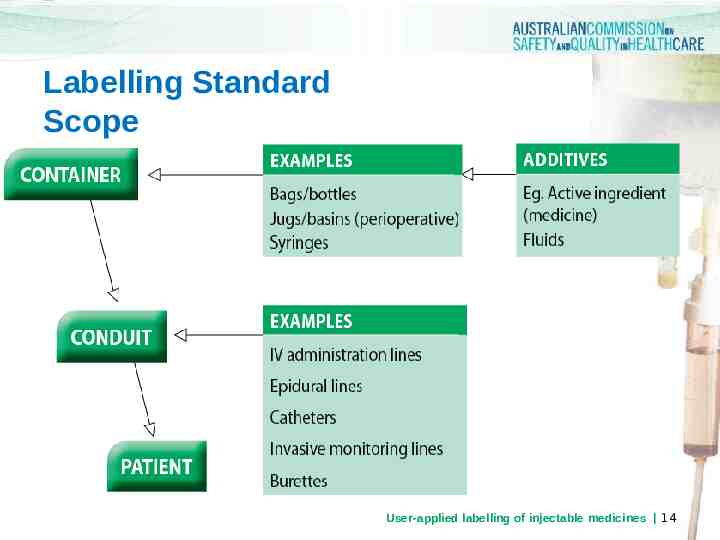
Labelling Standard Scope User-applied labelling of injectable medicines 14
Labelling Standard Scope all clinical areas where injectable medicines and fluids are administered all injectable medicines and fluids prepared in the ward or clinical area injectable medicines, defined as any sterile medicine intended for administration by bolus injection, perfusion or infusion by the following routes: intravenous, intramuscular, intrathecal, intra-arterial, subcutaneous, intradermal, intraventricular, epidural, intravesicular, intravitreal, intrapleural and intra-ocular. This list is not exhaustive, and other routes of injection should also be considered in the context of the Labelling Standard (e.g. intraosseous and intraperitoneal) 15 User-applied labelling of injectable medicines
Labelling Standard Extended Scope Labelling of containers in perioperative settings (including cardiac catheter and interventional radiology units). The Anaesthetic Labelling Standard (ISO 26825:2008) applies to syringes containing medicines used during anaesthesia Colour coded pre-printed medicine labels for use on dedicated continuous infusion lines Liquid medicines for oral, enteral and inhalational use Labelling of non-injectable medicines and fluids prepared in the same area as injectable medicines. User-applied labelling of injectable medicines 16
Labelling Standard Exclusions Injectable medicines and fluids: prepared by hospital pharmacy departments, external manufacturers or compounding centres not directly administered to the patient e.g. ampoules Administration portals Topical products prepared when injectable medicines are not present; however, the same principles of identification translate to topical use of medicines, solutions, chemicals extemporaneously dispensed radiopharmaceuticals and reagents User-applied labelling of injectable medicines 17
Application in clinical practice User-applied labelling of injectable medicines 18
All Containers: Label content Patient: Write the patient’s given name and family name Identifier (ID): This is the URN or MRN or other local unique identifier for the patient DOB Date of Birth (DOB): This is a third patient identifier on the label For each medicine added to the container specify: Medicine name (active ingredient) Amount (total added to the container), including units Volume (total volume of fluid in the container) in mL Concentration (units/mL) Diluent (syringes only) Date and time of preparation Signed by personnel preparing and checking medicine User-applied labelling of injectable medicines 19
All Containers: Label content (continued) Diluent - complete for all syringes ‘Date’ and ‘Time’ the medicine is prepared ‘Prepared by’ and ‘Checked by’ to be signed by responsible personnel DOB : 18/5/72 Example of intravenous bag additive label User-applied labelling of injectable medicines 20
All Containers: Label content (continued) DOB: 17/8/63 DOB: 17/8/63 Example of intramuscular route syringe label Example of subcutaneous route syringe label User-applied labelling of injectable medicines 21
Identifying target tissue/ route of administration A standard colour system is used to identify the target tissue/intended route of administration* *Modified from Australian Standard AS4940 User-applied labelling of injectable medicines 22
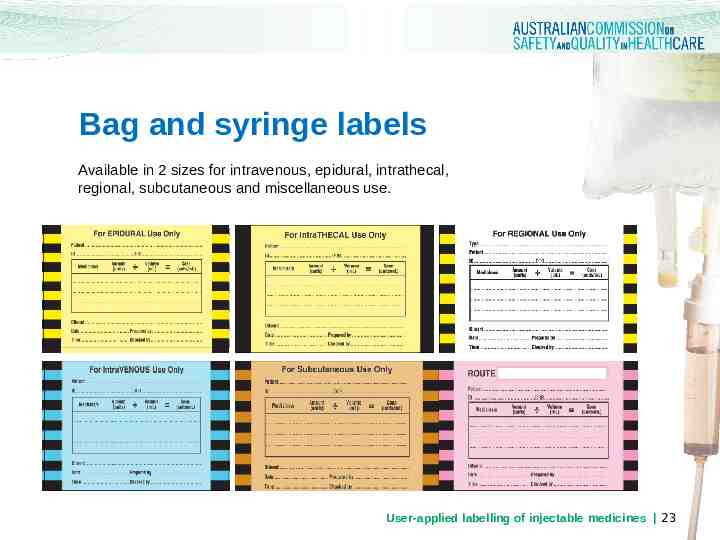
Bag and syringe labels Available in 2 sizes for intravenous, epidural, intrathecal, regional, subcutaneous and miscellaneous use. User-applied labelling of injectable medicines 23
Bags with additives Bags (and bottles) only require user-applied labels when a medicine is added in the clinical/ward area Label IMMEDIATELY an injectable medicine is added The ‘diluent’ should be identified on the label if the base fluid contained is not easily identifiable from the original manufacturers label (see label placement). User-applied labelling of injectable medicines 24
Bags with additives (continued) Placement: Place labels on the FRONT of the bag to ensure the name of base fluid, batch number and expiry date remain visible. DOB: 17/8/63 Place slightly off centre to ensure graduations on one side of the bag remain visible User-applied labelling of injectable medicines 25
Syringes For bolus or infusion Label all injectable medicines drawn up in syringes that leave the hand of the operator IMMEDIATELY. Prepare multiple syringes by preparing and labelling one syringe in an independent operation before preparing a subsequent medicine Labelling is NOT required when preparation and bolus administration of a SINGLE medicine from a SINGLE syringe are one uninterrupted process, and the syringe remains in the hand of the person who prepared it, and the same person administers the medicine IMMEDIATELY User-applied labelling of injectable medicines 26
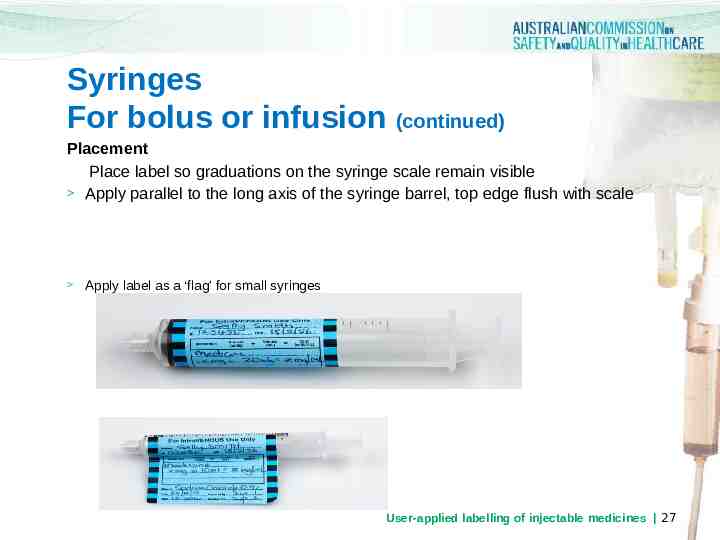
Syringes For bolus or infusion (continued) Placement Place label so graduations on the syringe scale remain visible Apply parallel to the long axis of the syringe barrel, top edge flush with scale Apply label as a ‘flag’ for small syringes User-applied labelling of injectable medicines 27
Labelling IV flushes Label any fluid drawn up in a syringe for use as an IV flush (e.g. 0.9% sodium chloride) unless preparation and bolus administration is one uninterrupted process Use an abbreviated preprinted 0.9% sodium chloride label Use full container labels for all other medicines and fluids User-applied labelling of injectable medicines 28
All containers: Discarding Content Any unlabelled container holding a solution must be immediately discarded Any container, where there is doubt over content, must be discarded Any medicine remaining in the container at the end of a procedure must be discarded User-applied labelling of injectable medicines 29
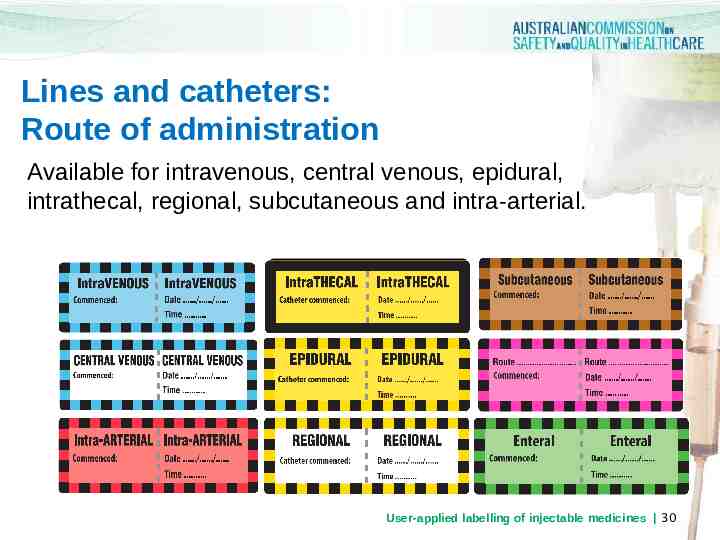
Lines and catheters: Route of administration Available for intravenous, central venous, epidural, intrathecal, regional, subcutaneous and intra-arterial. User-applied labelling of injectable medicines 30
Lines and catheters: Route of administration (continued) Labelling administration lines and catheters Label all lines to identify route Add date and time the line was commenced Identify catheters where there is a risk of wrong route administration, e.g. the patient entry portal is distant from the administration site Labelling invasive monitoring lines Identify all lines, including those not primarily intended for medicine administration. User-applied labelling of injectable medicines 31
Lines: Active ingredient Identify the medicine (active ingredient) within administration lines for dedicated continuous infusions Use preprinted labels where possible. Colour should comply with Anaesthetic Labelling Standard (and its extension) Lines for intermittent infusions do not need labelling for medicine. Any medicine label applied must be removed on completion of infusion User-applied labelling of injectable medicines 32
User-applied labelling of injectable medicines 33
Lines: Label Placement Route: Use colour coded route label Label near the injection port on the patient side *Exception where there is a possibility of tampering (e.g. paediatric patients) User-applied labelling of injectable medicines 34
Lines (continued) Label Placement Medicine (Active ingredient): Use pre-printed medicine label if available Use generic medicine label Label close to patient entry portal adjacent to route label *Exception where there is a possibility of tampering (e.g. paediatric patients) User-applied labelling of injectable medicines 35
Special circumstances No label required if: Preparation and bolus administration of a SINGLE medicine from a SINGLE syringe is one uninterrupted process -the syringe DOES NOT leave the hands of the person who prepared it, and - that same person administers the medicine IMMEDIATELY User-applied labelling of injectable medicines 36
Burettes User-applied labelling of injectable medicines 37
Burettes DOB: Use ‘peel-off’ labels reserved for use on burettes ONLY Place label so that text is upright and ensure that the burette graduations are not obscured Burette labels must be removed once the medicine has been administered to the patient User-applied labelling of injectable medicines 38
Catheter Lock User-applied labelling of injectable medicines 39
Catheter Lock For central venous access devices that are locked with a medicine (e.g. heparin) Label to partially cover the catheter dressing Remove label after removing medicine from the lock Label to have a ‘peel off’ adhesive strength to ensure dressing remains in place User-applied labelling of injectable medicines 40
Non-Injectable Medicine - ENTERAL ROUTE - INHALATION User-applied labelling of injectable medicines 41
Non-Injectable Medicine – Enteral Route Container and line labels available Syringes for non-injectable solutions must not be compatible with parenteral entry portals User-applied labelling of injectable medicines 42
Non-Injectable Medicine – INHalation Nebules are preferred source of solutions for inhalation If nebuliser solutions must be measured with a syringe then label the syringe User-applied labelling of injectable medicines 43
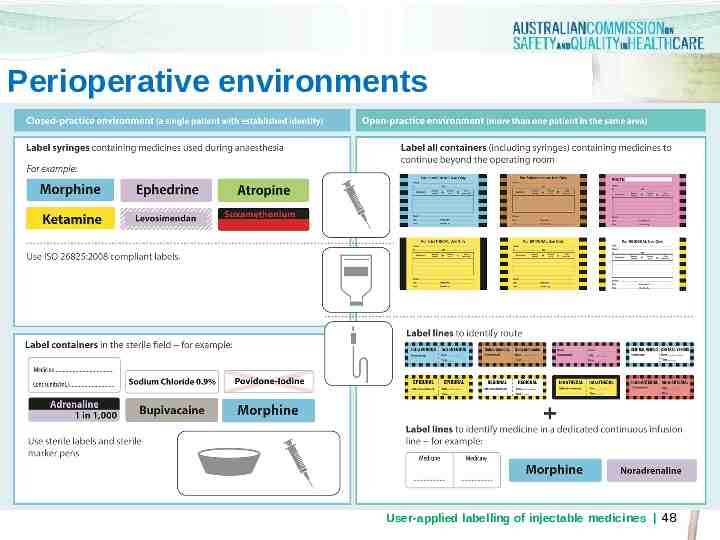
Closed –Practice Environments -Perioperative Sterile Field Sterile Field -Interventional Cardiology -Radiology User-applied labelling of injectable medicines 44
Sterile field (i.e. aseptic conditions) Closed-practice environment: where patient identification is established and other means of recording labelling and preparation signatories are available Any container holding medicines or fluids on the perioperative sterile field must be identifiable. Preprinted abbreviated container labels can be used Non-injectable medicines and fluids are identified with a red St Andrew’s Cross watermark Sterile markers must be available for use in the sterile field. User-applied labelling of injectable medicines 45
Perioperative environments Perioperative Environment User-applied labelling of injectable medicines 46
Perioperative environments Continue to label syringes containing drugs used during anaesthesia to comply with ISO26825:2008 Use preprinted labels or the ‘peel off’ abbreviated container label where patient identity is established and there are other means of recording labelling and preparation signatories User-applied labelling of injectable medicines 47
Perioperative environments User-applied labelling of injectable medicines 48
Perioperative sterile field Use preprinted label sheets with medicine name and concentration. Colour coding to follow ISO26825:2008 (Anaesthetic Labelling Standard) Use abbreviated container label where preprinted labels unavailable Labels must remain intact for duration of procedure Labels must adhere for duration of procedure Labels should be removed at the end of the procedure for reusable hollowware containers User-applied labelling of injectable medicines 49
Perioperative sterile field Example of preprinted label sheet for perioperative sterile field Note that labels for noninjectable fluids are clearly separated on the sheet User-applied labelling of injectable medicines 50
Interventional cardiology, radiology and other Perioperative Environment low-light procedure areas User-applied labelling of injectable medicines 51
Low-light procedure areas Use preprinted label sheets with medicine name Colour coding to follow ISO26825:2008 (Anaesthetic Labelling Standard) Example preprinted label sheet for cardiac catheter laboratory User-applied labelling of injectable medicines 52
Further information: Go to the Australian Commission on Safety and Quality in Health Care website www.safetyandquality.gov.au User-applied labelling of injectable medicines 53