MAT 272/BMSE 272 Mechanical Force and Biomolecules: Lecture 1
19 Slides3.64 MB
MAT 272/BMSE 272 Mechanical Force and Biomolecules: Lecture 1
Introduction: Why is force important for biomolecules?
1951: Pauling predicts alpha helix 1954: Watson and Crick propose double-helix Late 50s: Perutz, Kendrew attain first protein structures from X-ray crystallography 60s: Genetic code determined 70s: Amino acid sequences shown to uniquely determine protein function (Anfinsen) 80s: Biotechnology explodes (Molecular cloning , PCR) 90s: Genomes sequenced
A eucaryotic cell
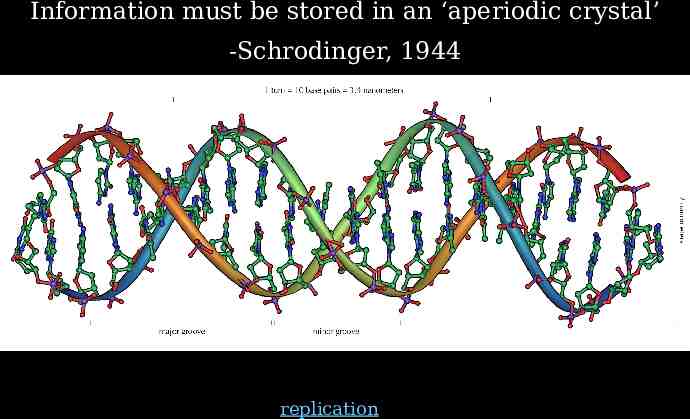
Information must be stored in an ‘aperiodic crystal’ -Schrodinger, 1944 replication
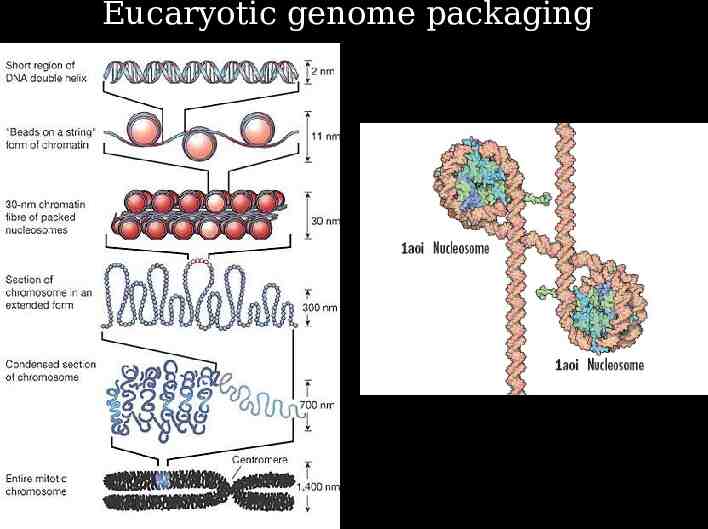
Eucaryotic genome packaging
Folded structures Proteins: Polypeptide chains that fold into globular structures with a wide variety of activities Myoglobin : one chain of 153 monomers; 17 kDa Proteasome: 28 chains of 200 monomers each; 6.8 MDa total A single-stranded RNA molecule can fold into an enzymatically active structure (a ribozyme)
1 3 4 2
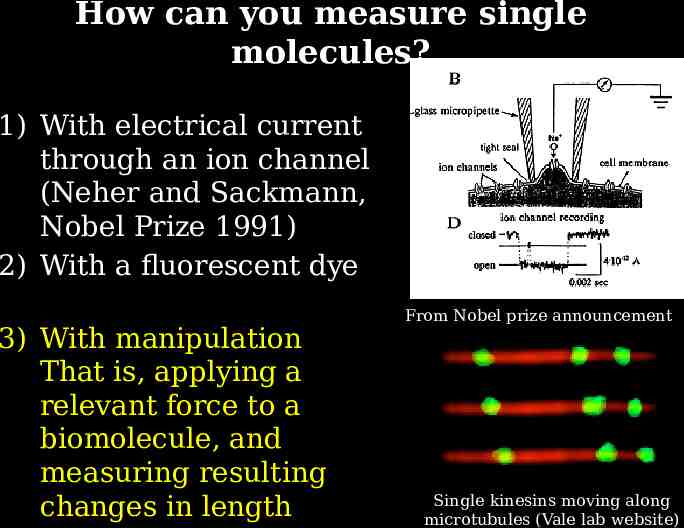
How can you measure single molecules? 1) With electrical current through an ion channel (Neher and Sackmann, Nobel Prize 1991) 2) With a fluorescent dye 3) With manipulation That is, applying a relevant force to a biomolecule, and measuring resulting changes in length From Nobel prize announcement Single kinesins moving along microtubules (Vale lab website)
The common techniques The Atomic Force Microscope The Optical Tweezer The Magnetic Tweezer The actuator A cantilever A dielectric bead A paramagnetic bead Position detection Quad photodiode Quad photodiode Video tracking Force range 10-1000 pN 1-200 pN 0.1-100 pN Advantages Bandwidth, sensitivity Bandwidth, manipulation Simplicity, constant force, rotation Disadvantag Limited low-force Low precision in
Units, and the interesting range of force Force Energy/Length; kBT 4 pN nm Lengt h Force Stretching DNA kBT 50 nm 0.1 pN Weak bonds kBT nm 4 pN Unzipping DNA 2/3 H bonds a few kBT nm 10-15 pN Motor motion ATP 20 k BT 1-10 nm 8-80 pN Covalent bonds [many weak nm bonds] 1 eV 40 0.1 nm k BT 10-200 pN 1 nN AFM Denaturing a protein Optical Energy tweezers Magnetic tweezers Process
Caveat: Reductionism
Interior of an E. coli cell; from Goodsell, 1991, by way of Alberts green: ribosomes; red: proteins; blue: Rna Certain proteins can only fold in the crowded interior of the cell; remove the crowding, and you’ve removed the physical impetus from the problem.
Anti-reductionism tracts 1) P. W. Anderson, Science (1972) “More is Different” This is the start of an argument that eventually killed the SSC. 2) C. R. Woese, Microbio. Mol. Bio. Rev. (2004) “A New Biology for a New Century” In which the author questions the significance of nearly the entire field of molecular biology.
“If you read trendy intellectual magazines, you may have noticed that ‘reductionism’ is one of those things, like sin, that is only mentioned by people who are against it. To call oneself a reductionist will sound, in some circles, a bit like admitting to eating babies. But, just as nobody actually eats babies, so nobody is really a reductionist in any sense worth being against.” – Richard Dawkins
Practical matters Evaluation -Two graded problem sets (30% of grade) -Half-lecture presentations (70%): These will be based on a recent paper from the field. You will get a list of papers to choose from. With my approval, you can use a paper not on the list. The rough plan: I give 16 lectures, students give last 2-3, plus during final exam week (if necc.) Prereq: Prior knowledge of stat. mech., not of bio Note 5/28 is a UCSB holiday.
The website http://www.engr.ucsb.edu/ saleh/#Teaching I have posted, and will continually update: 1. PPT slides from lectures (when used) 2. pdf of lecture notes 3. pdf of journal articles referenced (e.g. Woese and Anderson articles) Also, there are some links to online resources (e.g. textbooks, journal search engines) that could be useful for background research for your presentation
Practical Matters: The syllabus Schedule and textbooks