Laboratory Information Management Systems (LIMS) Lindy A. Brigham
18 Slides583.50 KB
Laboratory Information Management Systems (LIMS) Lindy A. Brigham Div of Plant Pathology and Microbiology Department of Plant Sciences PLS 595D Regulatory Issues in Laboratory Management February 2, 2006
LIMS Definition: – A method a laboratory uses to manage its data and disperse the results to designated areas
Information Technology Why? – Management of the Information Explosion – Quality Assurance GLP, GMP, EPA, etc. – Reduction of data entry errors – Need for faster sample/results turnaround time
General Considerations Different labs have different requirements Procedures and outcomes differ But all can use the systems approach to identifying their needs and implementing solutions
Types of Laboratories Research Analytical Services Manufacturing
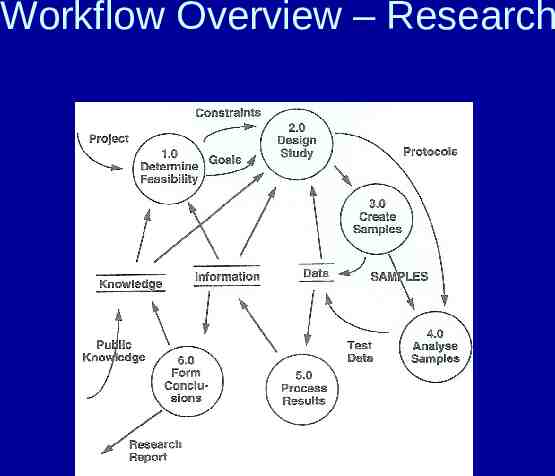
Research Lab Many non-routine tests Low sample volume Flexibility between data and analyses performed Specification checking (FDA reqs) Traceability (calibration, distribution, data) Change control procedures (documentation of changes eg SOPs, QA compliance)
Testing Services Laboratory Tests performed at customer’s request High sample volume Routine tests Variable workload Prioritized samples
Manufacturing Laboratory Perform testing for various materials including: – Final product testing – Stability testing – Work-in-progress testing – Environmental testing – Raw materials acceptance testing Many tests per sample Dynamic environment
LIMS Acquisition How do you determine what you need and how to get it? Systems Approach to defining needs and specifications for a LIMS
Acquisition Decisions Buy or Build? All or Pieces? Options – Design and build system ‘from scratch’ – Piece together existing and off the shelf components – Buy whole system from vendor How do you decide?
By doing an Analysis The build or buy or buy what decision is made based on an analysis of the needs and practices of the lab The analysis itself can be of benefit to any lab to: – Clarify existing practices – Help eliminate non-essential practices – Organize and streamline existing procedures
Systems Analysis and Design Project Definition – What is to be achieved, by whom and why Functional Requirements – all user entry requirements and system output requirements described in detail Functional Design – detailed documentation to describe the system and detail how the functional requirements are to be achieved – independent of hardware and software requirements Implementation Design – selection of hardware and software System Integration – Gather all required components, interface components, install software, go live System Evaluation – Final phase – Project definition and functional requirements are revisited and compared to the final installed system to determine how well the requirements were met
Analysis of UA Research Lab
Workflow Overview – Research
Workflow Overview – Analytical
Information Management System Consists of: – Computer(s) – People – Procedures/Processes
LIMS Resources Stand alone software – All purpose Word Processing Spreadsheets Databases – Inventory – Document Management (Wolk) – Statistical Packages Total systems – Lablynx – Starlims http://www.lablynx.com/ http://www.starlims.com/
Regulatory Requirements ISO 9000 – Management must define, implement, communicate and maintain quality objectives and assign personnel at all levels of the organization to be responsible for verifying the company’s quality system – Primarily effect manufacturing laboratories ISO 25 – Establishes labs technical competence GALP (Good Automated Laboratory Practices) – Union of federal regulations, policies, and guidance documents establishing a uniform set of procedures to ensure the reliability and credibility of laboratory data (EPA) 21 CFR 11 (1997) – Electronic signatures, Electronic records