IV Induction Agents Jed Wolpaw MD, M.Ed
59 Slides279.88 KB
IV Induction Agents Jed Wolpaw MD, M.Ed
History 1656 Percival Christopher Wren and Daniel Johann Major tried injecting wine and beer into a dog’s vein. Early 1900s Hedonal, ether, chloroform into veins—syncope, cyanosis, pulmonary edema Bier tried novacaine in veins—Bier block 1936 Thiopental Jarman 1946 &Miller’s Anesthesia 8th edition
Propofol Propofol a) Mechanism of Action b) Pharmacokinetics and Pharmacodynamics c) Metabolism and Excretion d) Effect on Circulation e) Effect on Respiration f) Effect on Other Organs g) Side Effects and Toxicity h) Indications and Contraindications
Propofol History Introduced in 1970s, now most widely used IV hypnotic Developed in UK, initially 1977 caused anaphylaxis Relaunched as emulsion in soybean oil in 1986
Mechanism of Action Alkylphenol derivative Enhances GABA-induced chloride currents through binding to B subunit of GABA-A receptor. At lower concentrations has indirect effect-potentiates activation by GABA At higher concentrations activates GABA-A directly Also inhibits NMDA glutamate receptors Increases dopamine in nucleus accumbens (sense of well-being) Decreases serotonin in area postrema (anti-emetic)
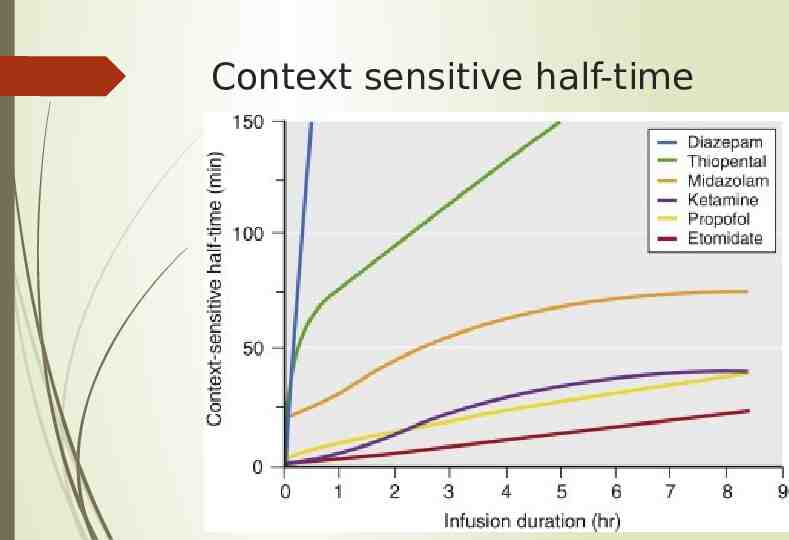
Pharmacokinetics Oxidized and conjugated in liver and then excreted by kidneys There is some extra-hepatic clearance likely in kidneys and lungs Competitive inhibitor of CYP3A4, increases duration of action of midazolam After single bolus dose levels decrease rapidly due to redistribution and elimination with initial distribution half-life 2-8min Context sensitive half-time 10 minutes for infusions up to 3 hours and 40 minutes for 3-8hours. Starts to reduce EEG activity in 20 seconds, peak effect in 90 seconds Because of decreased cardiac output and therefore decreased hepatic blood flow, can impair its own clearance
Pharmacokinetics Greater reduction in cardiac output in older patients 80 y/o needs 50% the dose of a 20 y/o to get same effect Even though it is cleared hepatically no dose reduction needed in hepatic disease due to efficient clearance and extra-hepatic clearance Increases plasma levels of midazolam and remifentanil
Context sensitive half-time
Pharmacodynamics Onset of hypnosis after dose of 2.5mg/kg is 20 seconds with peak effect 90 seconds ED50 for loss of consciousness is 1-1.5mg/kg Duration of action is dose dependent, after 2mg/kg lasts 5-8 min Highest dose needed in less than 2 y/o Can suppress seizures but can also cause them Decreases ICP by 30-50% and decreases CPP, decreases intraocular pressure by 30-40% Cerebral autoregulation relatively intact
Effects on respiration Induction dose produces apnea for 30 seconds or longer with other medications Maintenance infusion decrease tidal volume by 40% and increases RR by 20% even up to 200mcg/kg/min May have some bronchodilatory properties
Effects on cardiovascular system Decreased BP by 25-40% due to decreased CO and SVR HR can go up or down by about 10%-inhibits baroreceptor reflex tachycardia that would normally results from hypotension Hemodynamic effect lags behind hypnotic effect (twice as long) Can suppress atrial tachycardias-avoid in EP studies Reduces myocardial blood flow and O2 consumption preserving supply to demand ratio May have some cardioprotective effects at higher doses and in combination with inhaled anesthetics
Use Induction Maintenance 25-75mcg/kg/min Anti-emetic 50-150mcg/kg/min based on total body weight, adjusted to individual (older and sicker patients require much less) Sedation 1-2.5mg/kg based on lean body mass, adjusted for age, reduced with premed 10-20mg IV intermittently or infusion of 10mcg/kg/min Infusions 30mcg/kg/min usually cause amnesia
Advantages Does not prolong neuromuscular blockade but large doses may provide good intubating conditions without paralysis Not an MH trigger No adrenal suppression Pleasant dreams Anti-emetic even at low doses (10mcg/kg/min) When used as maintenance in breast surgery was more effective than 4mg Zofran at preventing PONV Also relieves pruritis from opiates and from cholestasis
Adverse effects Anaphylactoid reactions, especially in people with multiple allergies Pancreatitis-likely from hypertriglyceridemia-older, longer duration, higher dose Pain on injection-rare thrombophlebitis of vein Propofol infusion syndrome Usually doses 70mcg/kg/min for 48h Acute bradycardia leading to asystole with metabolic acidisos, rhabdo, hyperlipidemia, fatty liver, hyperkalemia Likely a genetic disorder involving fatty acid metabolism is involved
Barbiturates a) Mechanism of Action b) Pharmacokinetics and Pharmacodynamics c) Metabolism and Excretion d) Effect on Circulation e) Effect on Respiration f) Effect on Other Organs g) Side Effects and Toxicity h) Indications and Contraindications
Uses Thiopental: Induction, cerebral protection (e.g. status epilepticus) Methohexital: Induction, especially for ECT Phenobarbital: Seizure suppression
Mechanism of Action Bind to GABA-A receptor and enhance GABA effect (low doses) and directly stimulate (high doses) causing increased chloride current and hyperpolarization Reduces CMRO2, ICP and CBF but preserves CPP due to greater drop in ICP than MAP
Pharmacokinetics All hepatically metabolized then excreted in urine except phenobarbital (60-90% renal unchanged) Rapid redistribution terminates action of single dose Much longer context sensitive half-time than propofol
Pharmacodynamics Single dose lasts 5-10 minutes, redistributes to lean tissues Wake up is faster after methohexital than thiopental due to greater hepatic uptake Longer recovery of barbiturates in general led to replacement by propofol
Respiratory and CV Effects Dose dependent depression, apnea after induction dose Peripheral vasodilation and negative inotropy Increased HR 10-36% from baroreceptor reflex, can be harmful in cardiac disease Can prolong QT interval In hypovolemia can reduce CO by 70%
Adverse effects Garlic or onion tastes Allergic reactions Local tissue irritation or rarely necrosis Induction of P450 system Bronchoconstriction in asthmatics
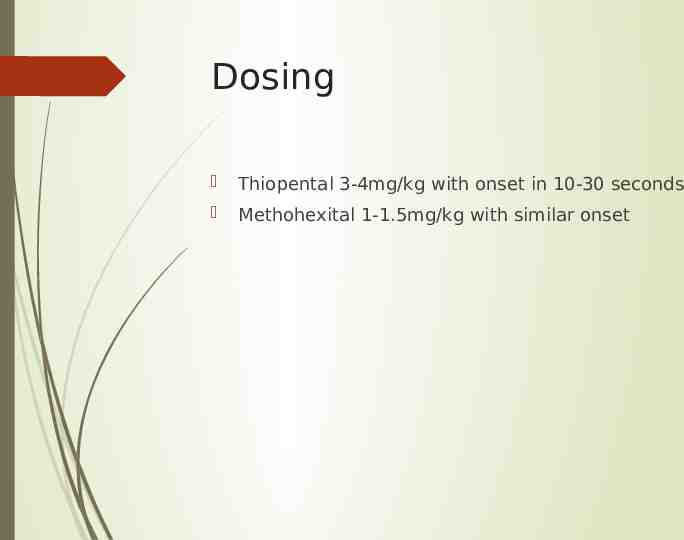
Dosing Thiopental 3-4mg/kg with onset in 10-30 seconds Methohexital 1-1.5mg/kg with similar onset
Benzodiazepines Midazolam (short acting) Lorazepam and Temazepam (intermediate) Diazepam (long acting) Flumazenil (antidote, shorter half life than most of the benzos)
Mechanism of Action Bind to GABA-A receptor, enhance response to GABA Leading to hypnotic, sedative, anxiolytic, amnestic, anticonvulsant, muscle-relaxation properties Increase seizure threshold Maintain normal CBF to CMRO2
Pharmacokinetics Midazolam Rapid onset 1min, peak 2-3 min Distribution half life 6-15 min Hepatically metabolized by CYP system including active metabolite 1hydroxymidazolam Not an issue with normal renal function but in renal impairment can build up Lorazepam Conjugated in liver (not by CYP) to inactive compounds so renal impairment doesn’t prolong action but liver failure can Infusion can cause propylene glycol toxicity Remimazolam New, cleared by non-specific tissue esterases
Effects on respiratory system Reduce muscular tone in upper airway leading to risk for obstruction Reduce response to increased CO2 Reduce hypoxic response Synergistic effect with opioids Midazolam PO has little effect on respiration
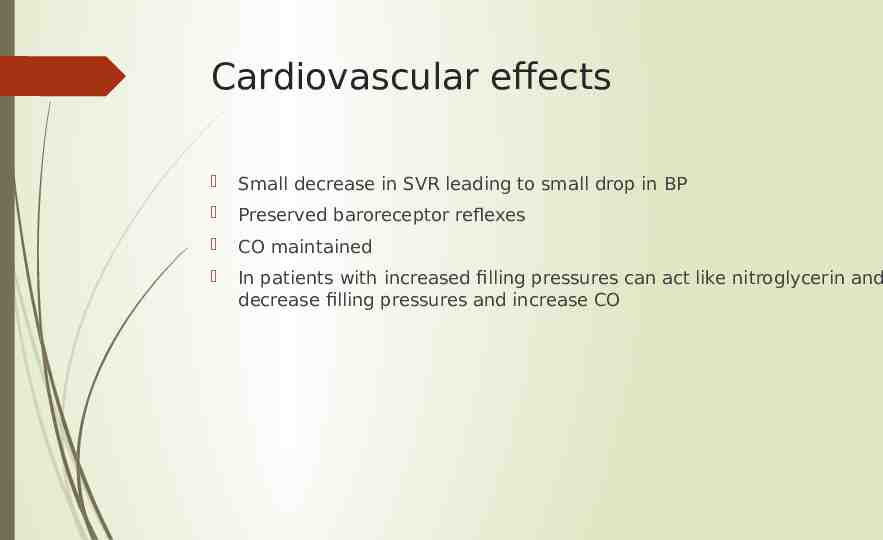
Cardiovascular effects Small decrease in SVR leading to small drop in BP Preserved baroreceptor reflexes CO maintained In patients with increased filling pressures can act like nitroglycerin and decrease filling pressures and increase CO
Uses and doses Premedication: anxiolysis, amnesia, reduced PONV Midazolam 7.5-15mg PO or 0.5-2mg IV Lorazepam 2-4mg PO or 0.25-1mg IV ICU sedation Induction of anesthesia Avoid if possible due to delirium, prolonged action Midazolam 0.3mg/kg adjusted for age Reduction of PONV IV doses as low as 1mg after induction
Adverse effects Lorazepam and Diazepam can cause venous irritation and thrombophlebitis All can have extended effects Post-operative delirium Give appearance of sleep without restorative sleep
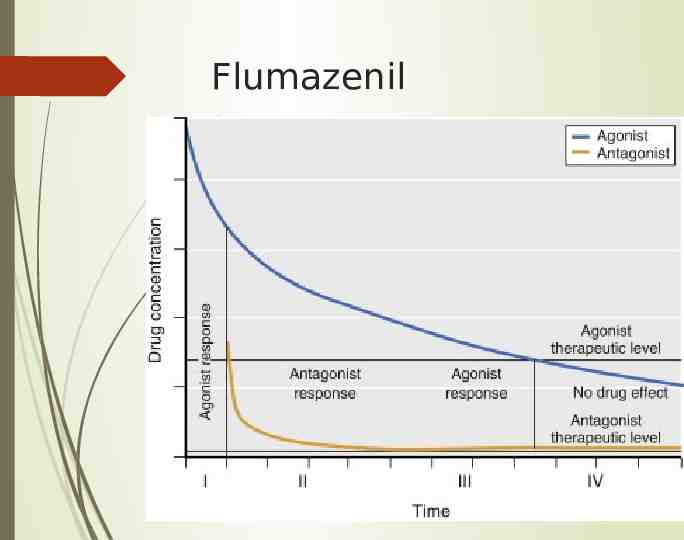
Flumazenil Competitive antagonist Short half-life, may get rebound effect of agonist, can use infusion (3060mcg/min) Rapid onset, 1-3 min, lasts 3-30 min Can cause seizures in patients on chronic benzos
Flumazenil
KETAMINE Synthesized in 1962, first used in humans in 1965, released for clinical use 1970 Phencyclidine-bines to NMDA Receptor Racemic mixture of S and R ketamine. S isomer is more potent and has fewer psychomimetic effects Only 20% bioavailability after oral use 2/2 first pass metabolism
Mechanism of action Acts at NMDA receptors, opioid receptors and monoaminergic receptors Most important is inhibition of NMDA glutamine input to GABA system centrally as well as in spinal cord where it also inhibits ACh release Produced “dissociative anesthesia” because patients may not appear asleep (eyes open, reflexes intact)
Pharmacokinetics Metabolized in liver to norketamine and hydroxynorketamine Norketamine has some activity but less than ketamine Metabolites excreted in urine Bioavailability orally is 20-30%, intranasally is 40-50%
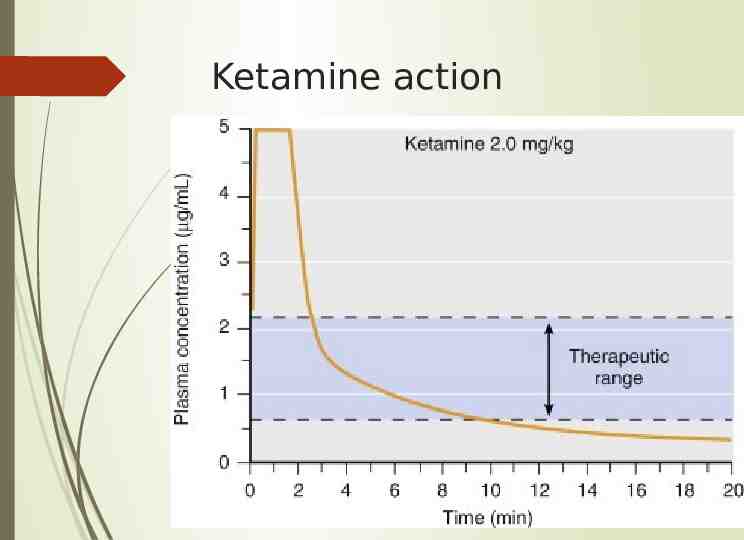
Ketamine action
Pharmacodynamics Onset in 30-60 seconds, peak in 1 minute, duration about 8-10min for anesthetic properties, 15-30 min for full orientation Pupils dilate, nystagmus Lacrimation and salivation Increased muscle tone, purposeless movements Prolonged by benzos
Advantages for post-op analgesia Sub-anesthetic doses can produce analgesia Inhibits central hypersensitization Attenuates acute tolerance to opioids and opioid induced hyperalgesia
Effects on CNS Increases CMRO2, CBF and ICP Preserves responsiveness to CO2 Emergence reactions 10-30% after pure or partial ketamine anesthetic Vivid dreams, extracorporeal experiences, hallucinations Last 1 to several hours Can be reduced by benzo co-administration
Effect on Respiratory system Transient decrease in minute ventilation after bolus (2mg/kg) but rarely apnea Bronchial smooth muscle relaxant-improves compliance in asthma Can also directly treat status asthmaticus Increased salivation, can be modulated by atropine or glycopyrrolate
Cardiovascular effects Usually causes increased BP, HR and CO Direct cardiodepressant effect Indirect stimulant effect by activation of sympathetic nervous system causing release of catechols, inhibition of vagus, inhibition of NE reuptake Can cause pulmonary hypertension If presynaptic catechol stores are depleted, cardiac depression will dominate Propofol and benzos blunt hemodynamic effects such as tachycardia Bolus is more likely to cause than infusion
Uses Induction In hemodynamically unstable patients or bronchoconstriction Good in tamponade Pain management Part of multimodal regimen, reduces opiate use Chronic pain Management of acute procedures like fracture reduction Sedation Great in pediatrics, fewer emergence reactions
Doses Induction: 0.5-2mg/kg IV or 4-6mg/kg IM Maintenance 30-90mcg/kg/min Sedation 0.2-0.8 mg/kg of 2-3min or 2-4 mg/kg IM
Adverse effects Increased ICP and IOP Increased myocardial O2 consumption Repeated abuse can cause liver and renal toxicity Preservative, chlorobutanol, is neurotoxic, can’t be given neuraxially, but S-ketamine comes in preservative free formulation
ETOMIDATE First clinical use in 1972 Widespread use in 1970s Began to taper off in 1980s with reports of adrenal suppression
Mechanism of Action GABA-A facilitation (lower dose of GABA required to activate receptor) At higher doses can activate receptor independently
Pharmacokinetics Cleared by Liver by ester hydrolysis then excreted by kidneys and in bile, metabolites not active Initial offset is due to redistribution so liver disease doesn’t alter it Initial dose (0.3mg/kg) lasts 6-8 min Clearance not altered by hypovolemia Short context sensitive half-time (less than propofol) but limited by adrenal suppression
Effects on CNS Reduces CBF by 34% and CMRO2 by 45% with no change in MAP CPP maintained or increased with improved cerebral O2 supply: demand Acutely decreases ICP by 50% if elevated (back to normal) but transient effect
Respiratory effects Induction dosing (0.3mg/kg) produces brief hyperventilation then brief apnea Ventilatory response to CO2 depressed
Cardiovascular effects Very hemodynamically stable even in hemorrhagic shock model Can see hypertension, tachycardia but less than ketamine Maintains myocardial O2 supply to demand ratio Has no analgesic effect so needs to be given with opiate to prevent HTN response to intubation
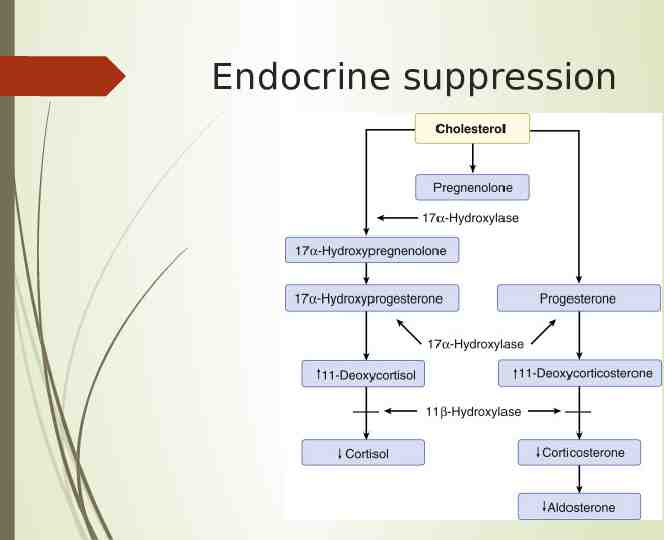
Endocrine effects Dose dependent inhibition of 11B-hydroxylase, decreased cortisol and mineralocorticoids Occurs at lower doses than hypnosis by more than 20x so suppression lasts much longer, up to 72h Can be used to treat hypercortisolemia
Endocrine suppression
Uses and Doses Induction (0.2-0.6mg/kg) Less if given with premed Good in trauma with potential hemorrhage or unstable hemodynamics Can produce longer seizures in ECT No longer used for prolonged sedation
Adverse effects High rate of nausea and vomiting Pain on injection Myoclonus (up to 70%), reduced by versed or magnesium prior Hiccups
Dexmedetomidine Alpha-2 receptor agonist, 1600:1 compared to 220:1 alpha-2 to alpha-2 compared to clonidine Used for sedation, anxiolysis, withdrawal, delirium S-enantiomer of medetomidine-used in veterinary medicine
Pharmacokinetics Almost complete biotransformation in liver with P450 system involvement Clearance is impaired in liver failure but not renal impairment due to inactive metabolites Severe renal disease sedative effect may be stronger due to decreased protein binding Context sensitive half time 4 minute after 10 min infusion and 250 minutes after 8 hour infusion So tapering probably not needed
Effects on CNS Sedative hypnotic effect through action on alpha-2 receptors in brain and spinal cord Independent of GABA system More natural sleep patterns than other drugs May have reduced rates of delirium Suppresses pain transmission in spinal cord Reduces post-op narcotic requirements by 50% when on infusion Reduces MAC Can enhance neuraxial blockade like clonidine epidurally Reduces CBF but unclear effect on CMRO2, autoregulation preserved Overall effect on supply:demand unknown Doesn’t reduce evoked potentials or seizure activity so can use in neuromonitoring or seizure mapping
Effects on Cardiovascular system Bradycardia (30% reduction), Reduced CO up to 35% Can see initial increase in BP with bolus loading dose from blocking peripheral alpha receptors
Uses and doses ICU sedation: 0.2-1.5 mcg/kg/hour Premed: 0.3-0.7mcg/kg slowly to prevent bradycardia Good as adjunct for awake crani Opioid sparing effects good for bariatric patients Awake fiberoptic intubation: sedation without respiratory depression and some dry mouth make it ideal Has been used to treat withdrawal and delirium, unclear data Weaning from vent in ICU
Adverse effects Bradycardia, hypotension, prolonged duration