Diagnosis and Treatment of Parkinson’s Disease Jeff Bronstein, MD,
28 Slides1.42 MB
Diagnosis and Treatment of Parkinson’s Disease Jeff Bronstein, MD, PhD Professor of Neurology at UCLA Director of the SW PADRECC Director of UCLA Movement Disorders
Parkinson’s Disease 2nd most common neurodegenerative disorder – lifetime risk: 1 in 40-100 Age of onset – Common after 60 y/o – Young onset (20-50 y/o) 10-15% Men get it more often than women 5% Inherited 95% likely caused by genetic predisposition and environmental influences
Parkinsonism Tremor (rest) Rigidity Bradykinesia/akinesia Decreased facial expression Stooped posture Micrographia/hypophonia Postural instability
Not all Parkinsonians have Parkinson’s Disease Neurodegenerative Disorders Idiopathic Parkinson’s Disease Multiple System Atrophy Progressive Supranuclear Palsy/CBGD Diffuse Lewy Body Disease Secondary Parkinsonism Vascular Neuroleptics Normal Pressure Hydrocephalus
Differential Diagnosis Parkinsonism Plus Multiple System Atrophy postural instability Shy-Drager dysautonomia Striatal nigral degeneration non dopa-responsive OPCA cerebellar dysfunction Progressive Supranuclear Palsy gaze paresis Diffuse Lewy Body Disease dementia Corticobasal Degeneration dystonia, apraxia Most do not respond to L-dopa and have early loss of postural reflexes
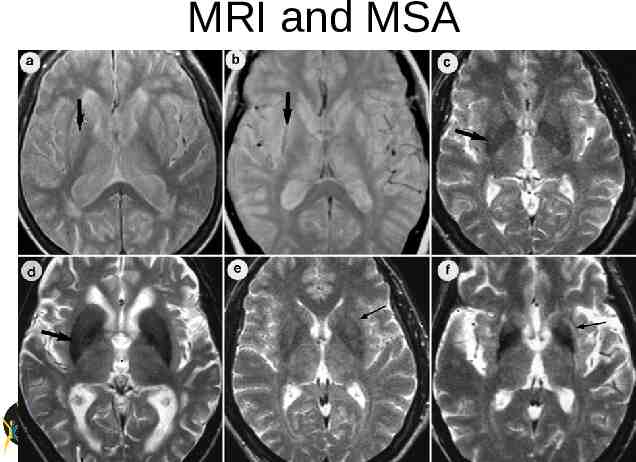
MRI and MSA
F-Dopa PET 18 QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Pavese and Brooks, 2008
Think Parkinson’s Disease With: Asymmetric onset L-dopa responsive Rest tremor Without: Cerebellar signs Long-tract signs Early dementia Early dysautonomia Early falls
Initiation of Treatment General Considerations – Age Young onset – neuroprotection – motor fluctuations Older patients – cognitive issues – comorbidities – Disability – Cost
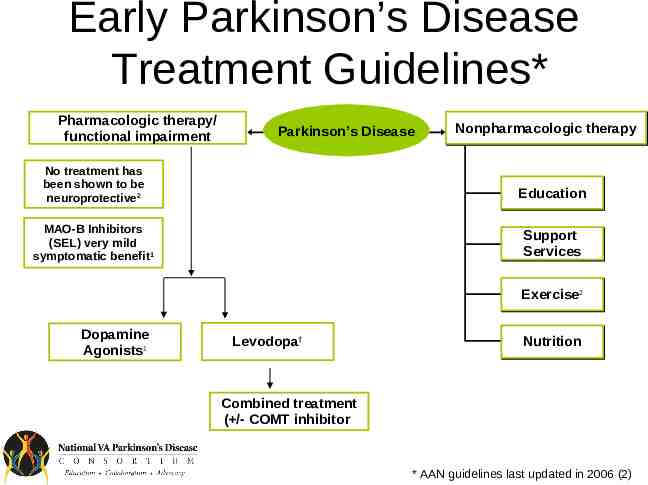
Early Parkinson’s Disease Treatment Guidelines* Pharmacologic therapy/ functional impairment Parkinson’s Disease Nonpharmacologic therapy No treatment has been shown to be neuroprotective2 Education MAO-B Inhibitors (SEL) very mild symptomatic benefit1 Support Services Exercise22 Dopamine Agonists1 Levodopa† Nutrition Combined treatment ( /- COMT inhibitor) * AAN guidelines last updated in 2006 (2)
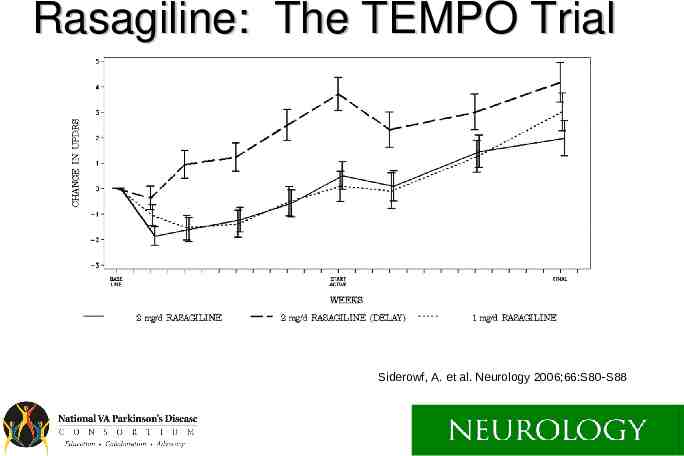
MAO-B Inhibition Selegiline and Rasagiline Both have small symptomatic effect. Both might slow disease down a little. Rasagiline and SL selegiline have been shown to help wearing off (PO selegiline not well studied).
Rasagiline: The TEMPO Trial Siderowf, A. et al. Neurology 2006;66:S80-S88
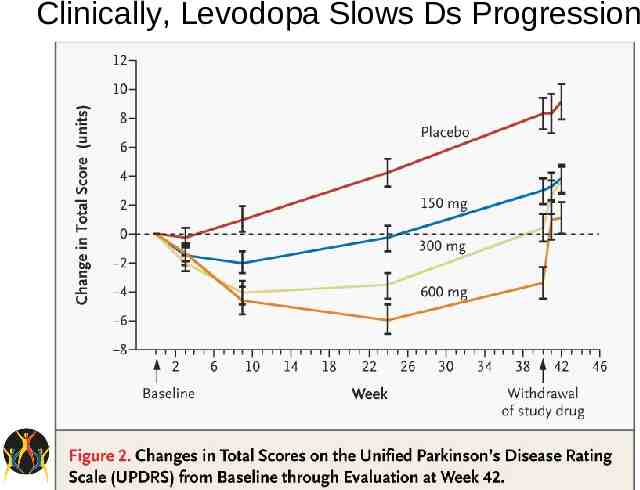
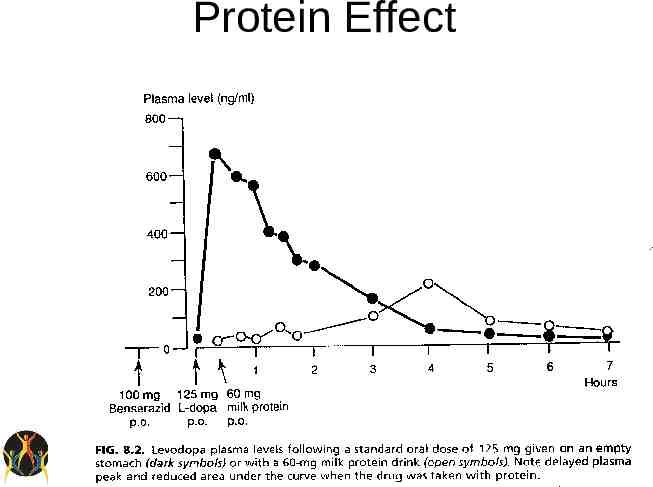
Levodopa Efficacy – Most efficacious medication for control of PD symptoms. – Improves UPDRS motor scores by approx 50% in advanced patients. – Short half-life – Significant protein effect Side-effects – Long-term risk of motor fluctuations
Clinically, Levodopa Slows Ds Progression
Protein Effect
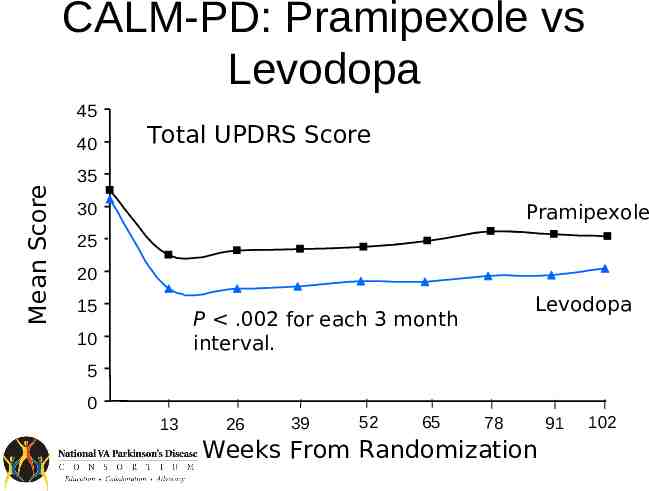
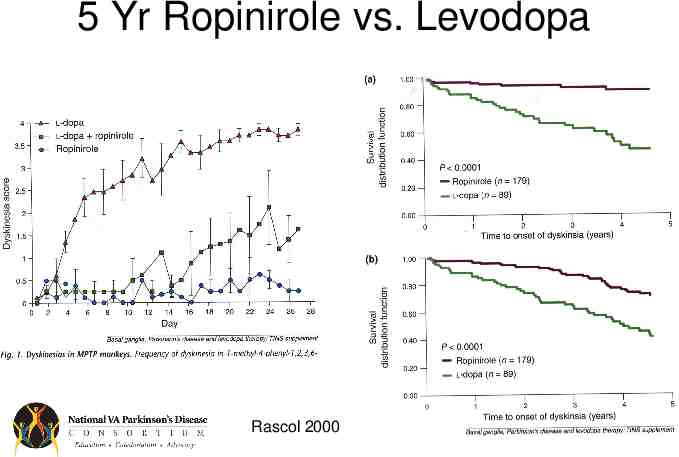
Dopamine Agonists Efficacy – – – – – Less efficacious than levodopa Have long half-lives Less likely to cause motor fluctuations Absorption without transporter (no protein effect) Potential alternate routes of administration (e.g. patch, injection) Side-effects – Relatively more common than for levodopa especially in the elderly – Include sedation, hallucinations, impulse control, nausea
CALM-PD: Pramipexole vs Levodopa 45 Mean Score 40 Total UPDRS Score 35 30 Pramipexole 25 20 15 Levodopa P .002 for each 3 month interval. 10 5 0 13 26 39 52 65 78 Weeks From Randomization 91 102
5 Yr Ropinirole vs. Levodopa Rascol 2000
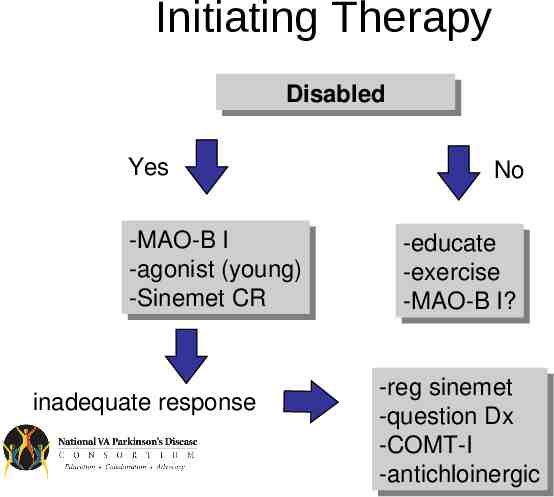
Initiating Therapy Disabled Disabled Yes -MAO-B -MAO-BII -agonist -agonist(young) (young) -Sinemet -SinemetCR CR inadequate response No -educate -educate -exercise -exercise -MAO-B -MAO-BI? I? -reg -regsinemet sinemet -question -questionDx Dx -COMT-I -COMT-I -antichloinergic -antichloinergic
Advancing Parkinson’s Disease Motor fluctuations (young) – Wearing off – Dyskinesias – On-off phenomenon Non-Motor Problems – – – – – Medication-induced psychosis Cognitive decline Postural instability Urinary problems Sleep problems
Principles of Managing Fluctuations Decrease fluctuations of L-dopa blood levels Use smaller more frequent dosing. Use combination of regular and CR Sinemet. Add COMT inhibitor Add MAO-B inhibitor Add DA agonist and reduce L-dopa Add amantadine for dyskinesias Surgery
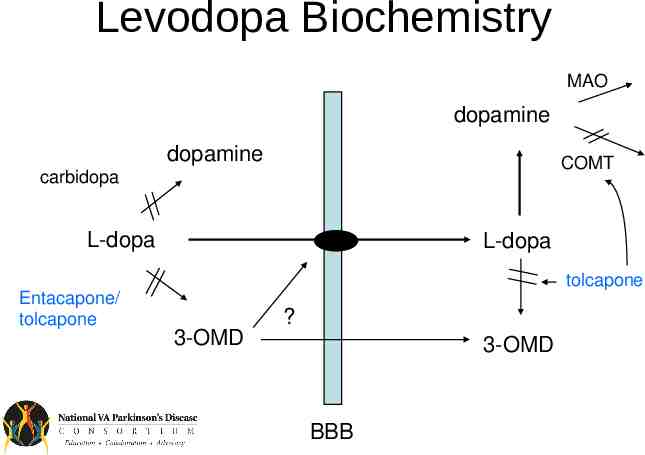
Levodopa Biochemistry MAO dopamine dopamine COMT carbidopa L-dopa L-dopa tolcapone Entacapone/ tolcapone 3-OMD ? 3-OMD BBB
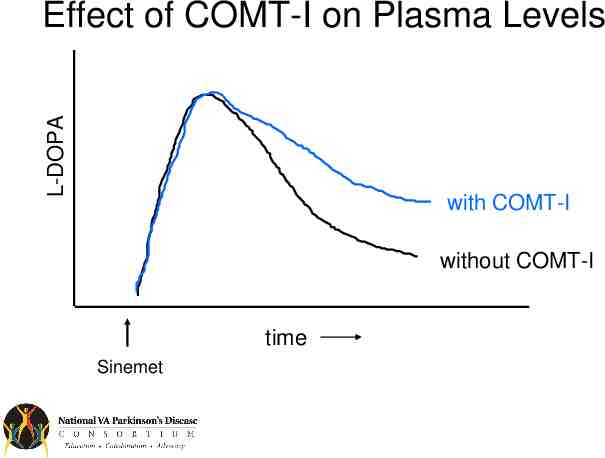
L-DOPA Effect of COMT-I on Plasma Levels with COMT-I without COMT-I time Sinemet
Advancing Parkinson’s Ds Hallucinations D/C selegiline, anticholinergics, amantadine lower dopaminergic medications (agonist 1st) clozapine, quetiapine, cholinesterase-I Falls optimize therapy R/O orthostatic hypotension physical therapy for training and assistive devices.
Advancing Parkinson’s Ds (cont.) Depression serotonin uptake inhibitor (e.g.Paxil, Celexa), nortriptyline, NA/Serotinergic uptake inhibitors, Wellbutrin Dementia R/o other causes (metabolic, structural etc.) Reduce medications as much as possible Consider cholinesterase-I, memenatine
Advancing Parkinson’s Ds (cont.) Sleep Problems sleep hygiene optimize DA therapy treat depression Consider sleep study (apnea, RSB) Sleep initiation: short acting benzo (Ambien, Sonata), Rozerem. Sleep maintenance: Lunesta, Ambien CR, tricyclic antidepressant (nortriptyline, trazadone), Remeron, Benadryl
Summary Motor fluctuations are treatable but can require time and persistence Identify and treat non-motor problems, they can be very disabling When in doubt, call or refer to the National VA Parkinson’s Disease Consortium http://www.vapdconsortium.org
Consortium Center Network Red Star – PADRECC Blue Star – Consortium Center Cyan Star-SW PADRECC